More Information
Submitted: January 27, 2026 | Accepted: February 02, 2026 | Published: February 03, 2026
Citation: Fredy NNG, Bervalle GKD. Pathophysiological Characteristics of Myelodysplastic Syndromes in Patients Residing in Brazzaville, Republic of Congo. J Hematol Clin Res. 2026; 10(1): 008-015. Available from:
https://dx.doi.org/10.29328/journal.jhcr.1008040
DOI: 10.29328/journal.jhcr.1008040
Copyright license: © 2026 Fredy NNG, et al. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Keywords: Myelodysplastic syndromes; Vitamin B12
Pathophysiological Characteristics of Myelodysplastic Syndromes in Patients Residing in Brazzaville, Republic of Congo
Nsonde Ntandou Gelase Fredy1,2* and Govi Kikonda Divine Bervalle1
1Laboratory of Physiology and Physiomythology of Animals, Faculty of Sciences and Techniques, Marien University NGOUABI, Brazzaville, B.P. 69, Congo
2Laboratory of Biochemistry and Pharmacy, Faculty of Health Sciences, Marien University NGOUABI, Brazzaville, B.P. 69, Congo
*Corresponding author: Dr. Nsonde Ntandou Gelase Fredy, PhD, Gelase Fredy, CAMES lecturer, Laboratory of Physiology and Physiomythology of Animals, Faculty of Sciences and Techniques, Marien University NGOUABI, Brazzaville, B.P. 69, Congo, Email: [email protected]; [email protected]
Myelodysplastic syndromes (MDS) are rare hematological malignancies often affecting the elderly. Globally, this disease accounts for 13% of cancerous blood diseases. MDS is generated by bone marrow dysfunction characteristic of ineffective hematopoiesis and the disorder of exogenous factors, including vitamin B12. The objective of this work is to study the pathophysiological characteristics of myelodysplastic syndromes in patients living in Brazzaville. The calculation of the hospital frequency of MDS, the haemogram, the myelogram, the selective grouping of blood cells by the cell profiler software, as well as the dosage of Vitamin B12, are the methods that allowed the realization of this work. Thus, the hospital frequency of MDS in Brazzaville is 4, 92%; the pathophysiological characteristics of MDS are marked by the morphological appearance of blood cells which reveals mostly the presence of metamyelocytes with banded nuclei and pleicaryocytes; The excess or deficiency of Vitamin B12 observed in MDS, is not responsible for its unexpected occurrence, it is therefore appropriate to remember that MDS contribute to the deformation of immune cells and that VIT B12 has no close relationship with the development of MDS.
As a component of the body, the normal state and physiology of blood cells determine the proper functioning of the organism. However, some hematological disorders manifest as dysfunction of one or all blood cell lines, sometimes accompanied by a vitamin B12 deficiency. This is the case with myelodysplastic syndromes (MDS), which most often affect the elderly [1]. These rare diseases represent approximately 13% of all diagnoses of hematological malignancies and are the first hematological disorder in individuals over 65 years of age [1]. They predominantly affect men, with a male-to-female ratio of 1.8 [2].
The etiology of myelodysplastic syndromes (MDS) remains unclear. They can be primary or secondary, particularly following exposure to cancer treatments (15% - 20% of cases). Certain environmental factors, such as active smoking or occupational exposures (especially benzene), are implicated, as are some genetic factors [3].
Worldwide, MDS affects 2–4 cases per 100,000 inhabitants per year. In individuals aged 70–80 years, the incidence rises to 70 cases per 100,000 inhabitants per year [4]. The annual incidence is 3 cases per 100,000 inhabitants, and as high as 20 cases per 100,000 inhabitants for those over 70 years of age. Among myeloid hematological disorders, these conditions occupy the 3rd position after acute leukemias and myeloproliferative syndromes with an incidence of 1.24/100,000 inhabitants/year [4,5]. In Europe, myelodysplastic syndromes (MDS) primarily affect individuals with an average age of 70. The annual incidence is 3 cases per 100,000 inhabitants, rising to 20 per 100,000 inhabitants for those over 70 [2]. In Africa, the incidence of MDS appears to be lower than in Europe, at 0.07 per 100,000 inhabitants per year, according to a cooperative, multicenter study conducted over 11 years from January 1995 to December 2005 [6].
In Central Africa, in general, and particularly in Congo-Brazzaville, the literature shows no studies conducted on MDS; therefore, we are interested in focusing our research on this condition by studying its pathophysiological characteristics. Epidemiological data on myelodysplastic syndromes (MDS) in Central Africa, in general, and particularly in Congo-Brazzaville, to date, do not provide information on the pathophysiological characteristics of MDS. The bodies of patients with MDS are very often exposed to infections, bleeding, and anemia due to dysmyelopoiesis, which leads to the presence of immature or defective leukocytes, platelets, and erythrocytes. Furthermore, this damage could potentially cause vitamin B12 deficiency, as vitamin B12 is an exogenous factor acting on all cell lines and is necessary for DNA synthesis [7-10]. Given this damage to bone marrow cells and the probable alterations in exogenous factors, what cytological and physiological disturbances are observed in MDS? To answer this crucial question, we deemed it necessary to conduct a study. Its objective is to study the pathophysiological characteristics of myelodysplastic syndromes (MDS) in patients attending the Brazzaville University Hospital.
Study setting
The study took place at two (2) sites, the University Hospital Center of Brazzaville (CHU-B) in the Hematology department for patient registration and the National Reference Center for Sickle Cell Disease (C.N.R.Dr) for sample processing.
Study type and period
This is an analytical, case-control study conducted from October 3, 2020, to April 3, 2021, a period of six (6) months. This period includes the sampling phase and the biological analysis.
Study population
Patients with and monitored for myelodysplastic syndromes (“cases”) and apparently healthy individuals (“controls”).
Inclusion criteria
The following patients were included: those being monitored or diagnosed with MDS, aged 30 to 80 years, with an abnormal complete blood count (CBC) and bone marrow aspirate, and who had voluntarily agreed to participate in the study through informed consent. For the Control group, individuals meeting the following criteria were included: healthy and not being monitored for any disease, aged 30 to 80 years, and with a normal complete blood count (CBC).
Exclusion criteria
Patients were excluded if they met any of the following conditions:
- Previously selected for MDS but whose bone marrow aspirate suggested a hematological disorder other than MDS (e.g., leukemia, aplastic anemia).
- Patients aged under 30 or over 80 years.
- Patients with a normal complete blood count (CBC).
- Patients with MDS also have another significant hematologic disorder (e.g., sickle cell disease, thalassemia).
- Patients with comorbid conditions that could interfere with study assessments, such as active infections, autoimmune disorders, or severe liver/kidney disease.
- Patients under treatment that could modify hematologic parameters (e.g., chemotherapy, immunosuppressive therapy, recent blood transfusions).
- Pregnant or breastfeeding women.
- Patients voluntarily refusing or unable to provide informed consent.
Sampling and sample size
The study used simple random sampling. Patients physically present at the time of the survey, who consented and met the inclusion criteria, were systematically recruited. The sample consisted of eight (8) patients with MDS and nine (9) controls, for a total of 17 samples.
Data collection
Patients were recruited from the hematology department of the University Hospital of Brazzaville (CHU-B). An interview was conducted with each patient. The purpose of the study was explained, and their verbal and written consent to participate was obtained. The interviews took place in a consultation room. Patients were interviewed alone or accompanied by a family member. The language used was French or one of the national languages, Lingala or Kituba. The choice of language was based on the language usually used at home, allowing for the best possible collection of the necessary information. A questionnaire was used to collect this information, based on the patient’s reasons for consultation, clinical signs, risk factors, biological factors, and other relevant details. This questionnaire is included in the appendix. This information was then transcribed into an Excel file using a computer for statistical analysis.
Materials
The materials consisted of biological materials, equipment, and small laboratory supplies.
Biological materials
The biological materials specifically consisted of peripheral blood and bone marrow aspirate collected from the radial vein and sternum, respectively, at the C.N.R.Dr. The laboratory started in November 2021. These samples were then collected in tubes (EDTA and dry) and on microscope slides (Figure 1).
Figure 1: Photograph of the biological material, A: Blood contained in the EDTA tube; B: Blood contained in the dry tube; C: Slide containing the bone marrow aspirate.
Investigation methods
This involved investigating the frequency and risk factors of myelodysplastic syndromes (MDS) at the University Hospital of Brazzaville (CHU-B). This involved collecting samples for analysis and conducting thorough tests. For this purpose, two biological samples were obtained per patient (a peripheral blood sample and a bone marrow aspirate sample).
Determining the frequency of MDS at CHU-B
To determine the frequency of MDS cases, we used statistical data from patients hospitalized in the hematology department of CHU-B during our study, which covered the period from November 2020 to February 2021. This allowed us to establish a ratio between the total number of patients admitted to this department and the number of patients whose diagnosis revealed MDS [11].
Description of the morphological appearance of blood cells in MDS
The description of the morphological appearance of blood cells in MDS was carried out using the complete blood count (CBC), revealing the quantity of the different cell lines affected in MDS. Its principle is based on an automated technique that transforms particle volume into an electrical signal, enabling volumetric particle detection by impedance variation, using the computer software integrated into the analyzer. After registering the samples in the sysmex analyzer and homogenizing them in a blender, they underwent a complete blood count (CBC) to determine the complete blood count (CBC) of the different patients: the EDTA tube was fitted to the Sysmex needle; approximately 25 μL of whole blood was aspirated for each sample; the results were provided within 2 to 3 minutes after sample aspiration and then printed. The analyzer (Sysmex) measured the total cell volume and cell count.
Peripheral blood smears
After homogenizing the blood in the EDTA tube, the slides were identified. A thin drop of peripheral blood was placed on each slide to prepare the smears. After air drying, the smears were fixed with May-Grünwald stain using a Pasteur pipette. A few drops of demineralized water were added to rinse off the stain. The slides were stained with Giemsa, then rinsed with clean water 15 minutes later. After air drying, the smears were examined under a light microscope, first at low magnification (10 x objective), then at high magnification (100 x objective) with the addition of immersion oil.
Bone marrow aspirate
Revealing the quality of the different cell lines affected in MDS: Its principle consists of performing a qualitative analysis of blood cells (RBCs, WBCs, platelets) from the bone marrow using a bone marrow biopsy.
Performing the bone marrow aspirate: After psychological preparation, the patient was positioned supine on the examination table. The puncture site was disinfected. The trocar was inserted into the sternum, then the stylet was removed, and a 20 ml syringe was inserted to aspirate the bone marrow fluid. Finally, a sterile dressing was applied to the puncture site.
Smear preparation: The bone marrow aspirate collected in the 20 ml syringe was immediately spread onto several slides to prevent rapid coagulation. The slides were then air-dried and labeled. May-Grünwald-Giemsa staining was performed. The slides were first examined at low magnification (10 x objective) to assess cellularity, then at high magnification (100 x objective) with the addition of immersion oil.
Selective grouping of pathological blood cells in bone marrow: This involves grouping certain blood cells to describe their specific appearance in myelodysplastic syndromes (MDS) were done using Cell Profiler Software, processing of myelogram images using the Cell Profiler, separation of cells from the primary image using the pixel, delimitation of the target cytoplasm area, cytoplasm segmentation, nucleus extraction, and complete nucleus isolation.
Vitamin B12 assay: The serum collected in the wells and stored in a freezer at -20 °C for five (5) months was then thawed at room temperature on the benchtop for 6 hours. It was then placed in microwells and subsequently in the pre-calibrated Cobas E 411 instrument for vitamin B12 assay, with reference values of 200-500 pg/mL.
Data processing: Statistical analysis was performed using Microsoft Excel to establish the database, R software (Version 3.4.4) to perform statistical tests, Student’s t-test to compare the means of the measurements, GraphPad Prism 6 to generate the figures, and Cell Profiler to process the myelogram images. The values are presented as means ± standard error. The significance level was set at p < 0.05.
Hospital frequency of MDS at CHU-B
According to statistical results from the Clinical Hematology Department of the University Hospital of Brazzaville (CHU-B) in the Republic of Congo, the total number of patients admitted between November 2020 and March 2021 was 174, including 8 cases of myelodysplastic syndrome (MDS) [12,13]. We used this total figure to calculate the hospital frequency of MDS, which is 4.59%.
Table 1 illustrates the sociodemographic variation and the reason for consultation of these patients. Their average age was 51.2 ± 12.3 years, and asthenia was their most common reason for consultation.
| Table 1: Sociodemographic factors and reasons for consulting the MDS. | |||||
| Variable | MDS, N = 8 | Control, N = 8 | [All], N = 16 | p - value | N |
| Age, Mean±sd | 51.2±12.3 | 39.5±3.21 | 45.4±10.6 | 0.032 | 16 |
| ♀ sex | 3 (37.5%) | 4 (50.0%) | 7 (43.8%) | 1.000 | 16 |
| ♂ sex | 5 (62.5%) | 4 (50.0%) | 9 (56.2%) | ||
| Married | 4 (50.0%) | 5 (62.5%) | 9 (56.2%) | 1.000 | 16 |
| Common-law partner | 4 (50.0%) | 3 (37.5%) | 7 (43.8%) | <0.001 | 16 |
| Reasons for consultation Asthenia + Pain and Fever | 1 (12.5%) | 0 (0.00%) | 1 (6.25%) | ||
| Asthenia + Dizziness + Palpitations | 1 (12.5%) | 0 (0.00%) | 1 (6.25%) | ||
| Asthenia + Dyspnea | 1 (12.5%) | 0 (0.00%) | 1 (6.25%) | ||
| Asthenia + Fever | 1 (12.5%) | 0 (0.00%) | 1 (6.25%) | ||
| Asthenia + Persistent Fever | 1 (12.5%) | 0 (0.00%) | 1 (6.25%) | ||
| Asthenia + Hydrocele | 1 (12.5%) | 0 (0.00%) | 1 (6.25%) | ||
| Physical asthenia | 1 (12.5%) | 0 (0.00%) | 1 (6.25%) | ||
| Persistent Fever | 1 (12.5%) | 0 (0.00%) | 1 (6.25%) | ||
| Nothing | 0 (0.00%) | 8 (100%) | 8 (50.0%) | ||
| Alcohol consumption | 0.041 | 16 | |||
| No | 6 (75.0%) | 1 (12.5%) | 7 (43.8%) | ||
| Yes | 2 (25.0%) | 7 (87.5%) | 9 (56.2%) | ||
Morphological aspect of blood cells in MDS
Complete Blood Count (CBC): The results of the complete blood count, showing the variation in the quantity of blood cells in patients and controls, are represented in Figures 2-4, respectively, as well as in Table 2. These results clearly demonstrate the difference between the hemoglobin level, the white blood cell count, and the platelet count in our study sample (patients and controls).
| Table 2: Vitamin B12, Diagnosis, and Cell Lineages. | |||||
| Variables | MDS | Control | ALL | p-value | N |
| N = 8 | N = 8 | N = 16 | |||
| Clinical signs | <0.001 | 16 | |||
| Asymptomatic | 8 (100%) | 0 (0.00%) | 8 (50.0%) | ||
| None | 0 (0.00%) | 8 (100%) | 8 (50.0%) | ||
| Definitve diagnosis | <0.001 | 16 | |||
| MDS | 3 (37.5%) | 0 (0.00%) | 3 (18.8%) | ||
| MDS+Megaloblastic Anemia | 2 (25.0%) | 0 (0.00%) | 2 (12.5%) | ||
| MDS with AREB | 1 (12.5%) | 0 (0.00%) | 1 (6.25%) | ||
| SMD with Pancytopenia | 2 (25.0%) | 0 (0.00%) | 2 (12.5%) | ||
| Healthy Subject | 0 (0.00%) | 8 (100%) | 8 (50.0%) | ||
| Vitamin B12 dosage, average ± Sd | 726 ± 822 | 726 ± 356 | 726 ± 612 | 0.999 | 16 |
| Erythrocyte lineage, average ± Sd | 3.08 ± 1.03 | 4.92 ± 0.83 | 4.00 ± 1.31 | 0.002 | 16 |
| Thrombocyte lineage, average ± Sd | 146 ± 218 | 212 ± 49.4 | 179 ± 156 | 0.428 | 16 |
| Leukocyte lineage, average ± Sd | 3.19 ± 1.89 | 4.18 ± 0.75 | 3.68 ± 1.48 | 0.202 | 16 |
| PN Neutrophil PN, average ± Sd | 1.20 ± 1.08 | 1.94 ± 0.54 | 1.57 ± 0.91 | 0.117 | 16 |
| Eosinophil PN, average ± Sd | 0.11 ± 0.18 | 0.15 ± 0.14 | 0.13 ± 0.15 | 0.656 | 16 |
| Basophil PN, average ± Sd | 0.01 ± 0.01 | 0.02 ± 0.01 | 0.01 ± 0.01 | 0.045 | 16 |
| Lymphocyte, average ± Sd | 1.56 ± 0.97 | 1.71 ± 0.28 | 1.64 ± 0.69 | 0.692 | 16 |
| Monocyte, average ± Sd | 0.30 ± 0.33 | 0.36 ± 0.14 | 0.33 ± 0.25 | 0.633 | 16 |
| Hemoglobin, average ± Sd | 7.47 ± 1.42 | 13.1 ± 1.72 | 10.3 ± 3.27 | <0.001 | 16 |
| PN: polymorphonuclear; Sd: standard deviation | |||||
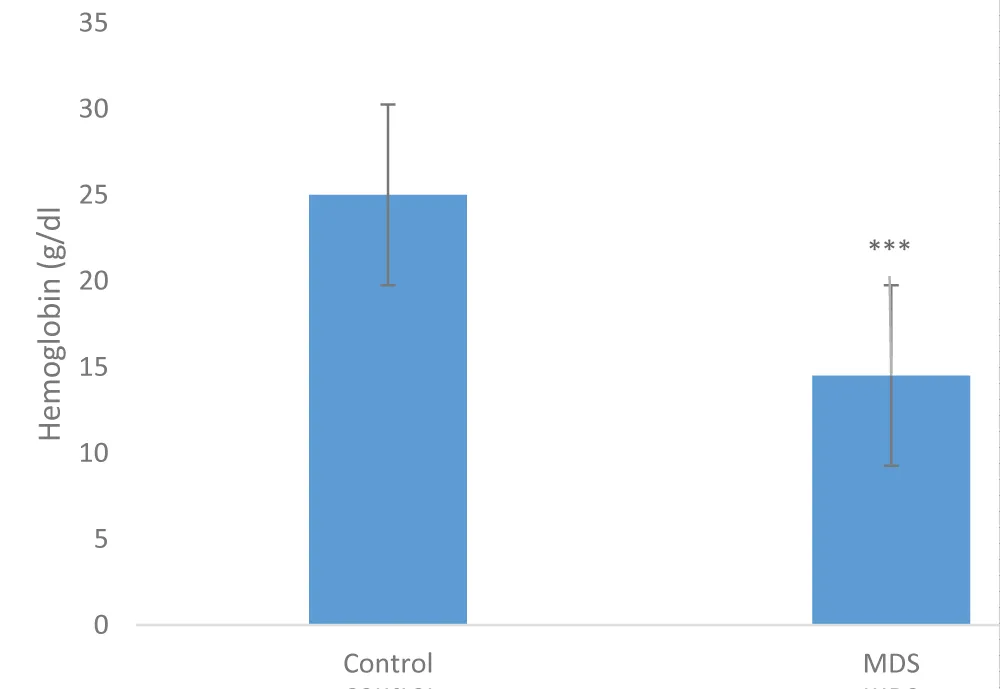
Hemoglobin level: The hemoglobin level in patients was very low compared to that of controls, with a highly significant difference (***p < 0.001), as shown in the distribution in Figure 2. This level ranged from 5.5 to 10 g/dL of blood in patients and from 13.5 to 15 g/dL in controls.
Figure 2: Hemoglobin distribution in patients and controls (***p < 0.001), n = 16.
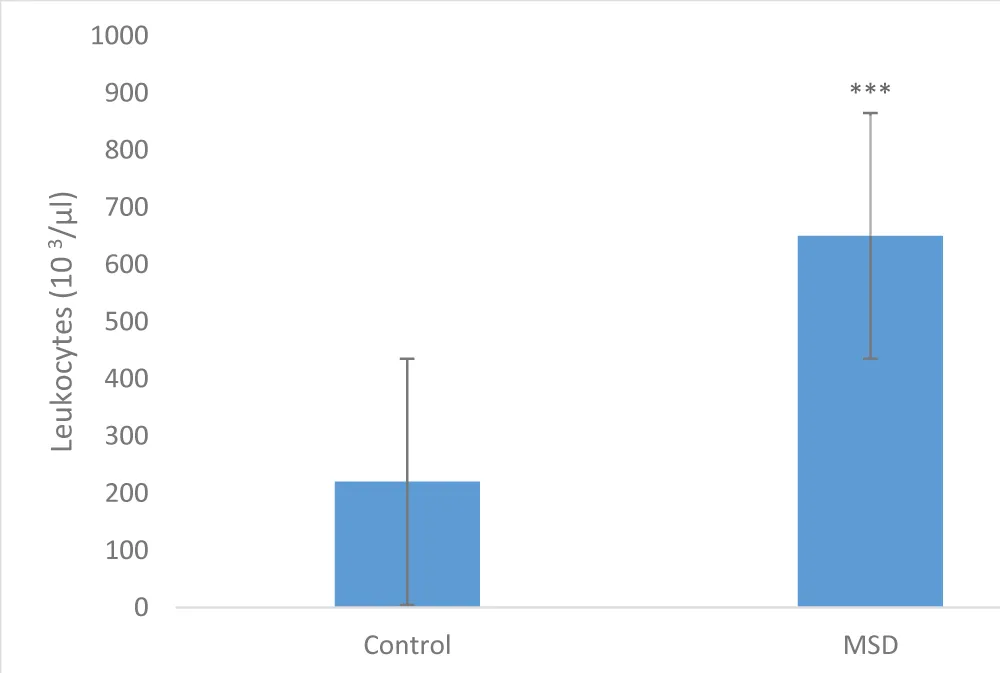
White blood cell counts: The distribution of white blood cell counts in patients and controls was unevenly distributed (p = 0.202), as illustrated in Figure 3. The distribution threshold in patients ranged from 1,000 to 6,000 µL and from 4,000 to 5,000 µL in controls.
Figure 3: Leukocyte lineage distribution in patients and controls (p = 0.202); N =16.
Platelet count
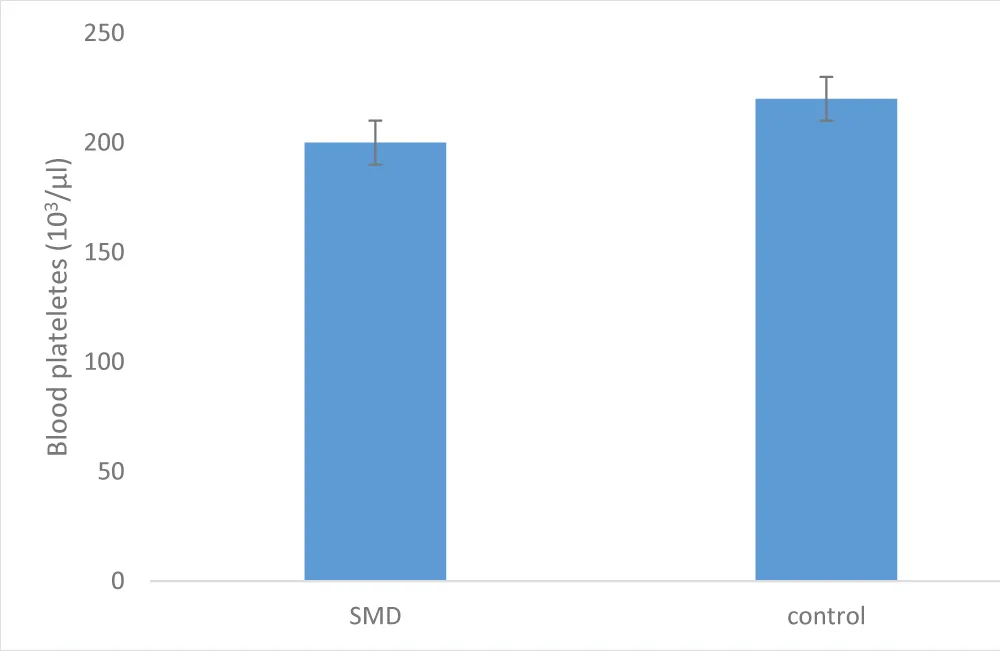
The distribution of platelet counts in patients and controls was unevenly distributed (p = 0.428), as illustrated in Figure 4. The distribution threshold for patients ranged from 1,000 to 6,500 µl and from 2,00 0 to 3,500 in controls.
Figure 4: Platelet lineage distribution in patients and controls (p = 0.428), N = 16.
Peripheral blood smears
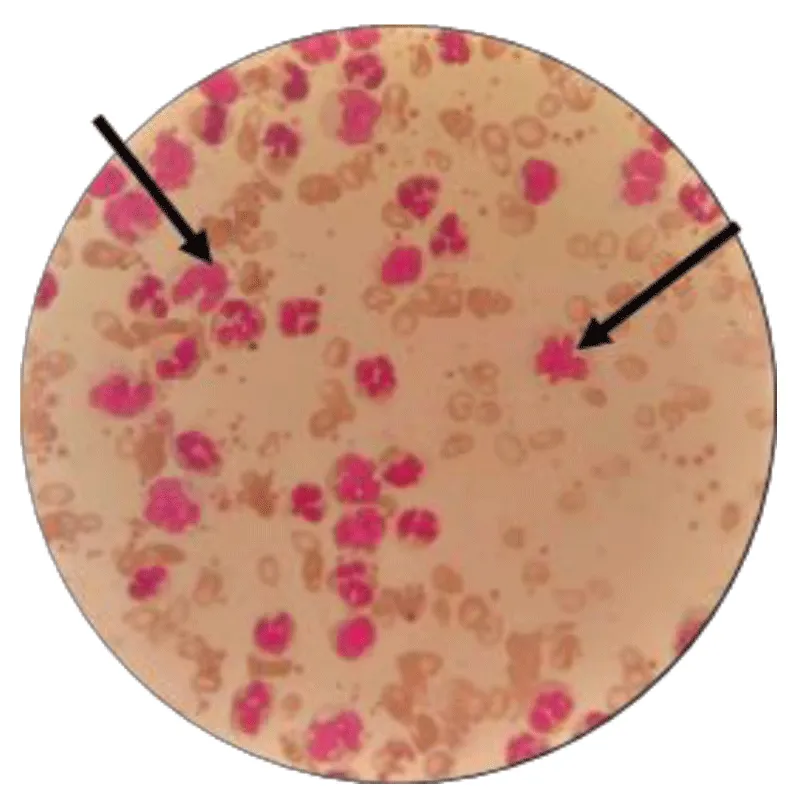
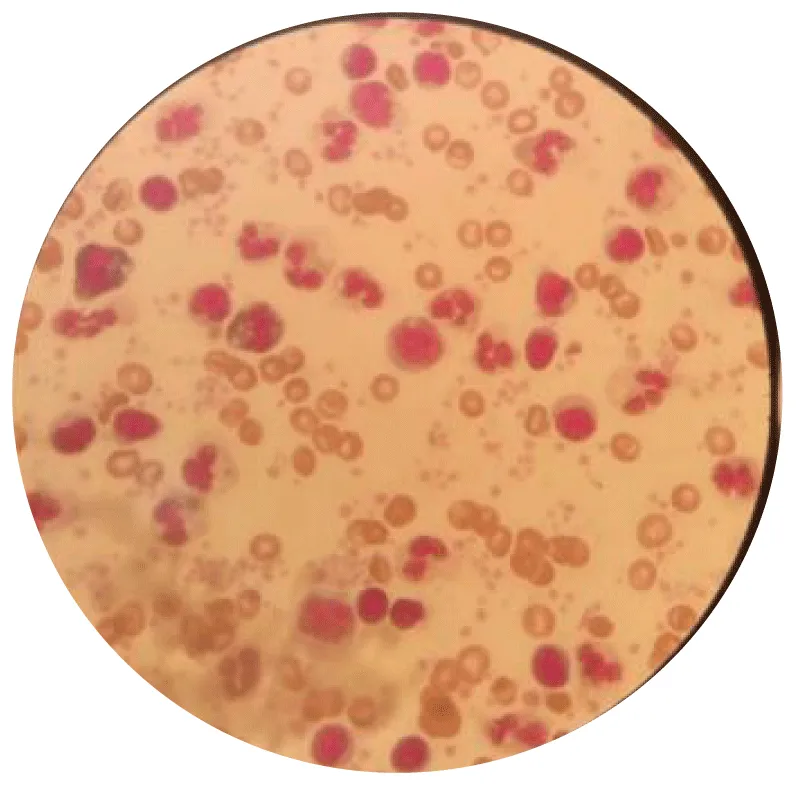
Images from peripheral blood smears performed on patients and controls revealed cellular malformations of certain lineages, particularly the myeloid lineage, as illustrated in Figure 5, in comparison with Figure 6, which shows a normal blood smear image from a control. In patients, the centrifugal arrows in the Figure 5 show the involvement of the myeloid lineage, respectively marked by the presence of a metamyelocyte with a ribbon-like nucleus and by that of a trefoil-shaped neutrophil. In controls, the Figure 6 shows the normal appearance of peripheral blood cells in a healthy subject, characterized by the presence of multilobulated neutrophils with normal-appearing cytoplasm and granulations surrounding the nucleus [14].
Figure 5: Appearance of peripheral blood cells during MDS (G x 100).
Figure 6: Appearance of peripheral blood cells in the control (G x 100).
Bone marrow aspiration
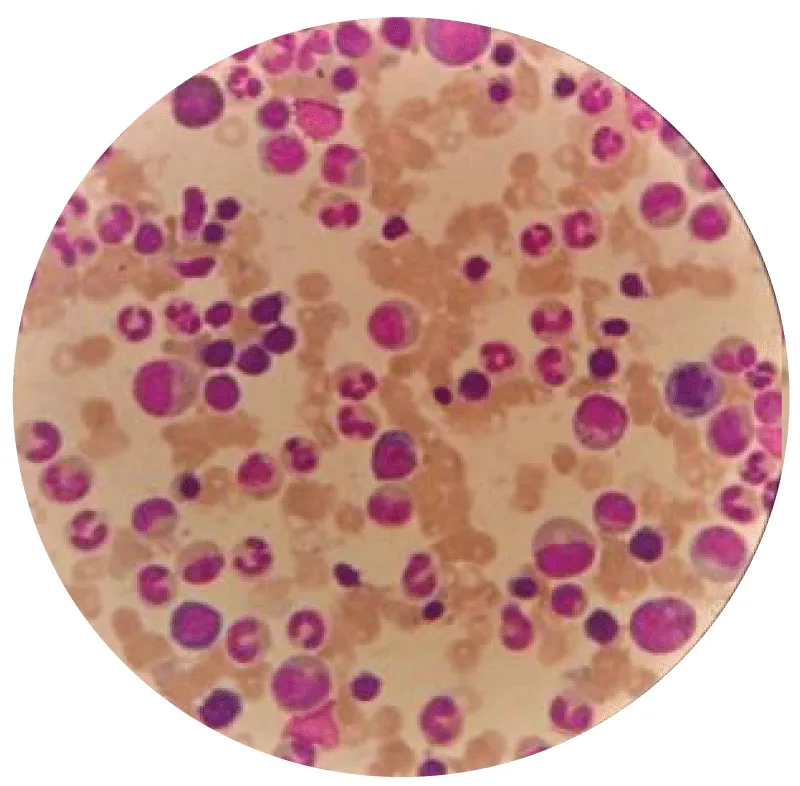
The bone marrow cells of patients with MDS had a very particular appearance during bone marrow aspiration, as illustrated in Figure 7. This appearance is marked by involvement of the myeloid lineage, resulting in the presence of numerous metamyelocytes with ribbon-like nuclei.
Figure 7: Appearance of bone marrow cells during MDS (G x 100).
A selective clustering of pathological bone marrow blood cells
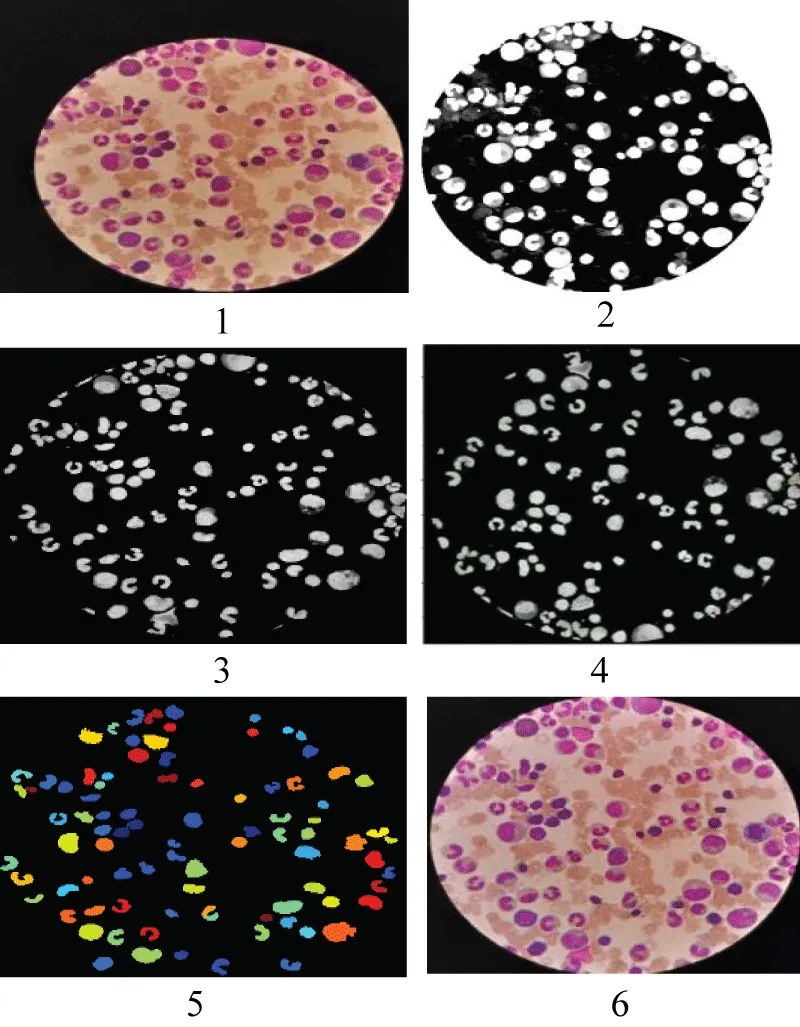
Figure 7 from MGm served as the original image during the exclusive processing of bone marrow blood cells by the Cell Profiler technique, generating steps 1, 2, and 3 as illustrated in Figures 8-10, marking the progressive, then complete, isolation of cell nuclei from MDS patients:
✓ Step 1: Separation and delimitation of cells from the primary MGm image using the pixel.
The Figure 8 explicitly illustrates the transformations of the primary MGm image, leading progressively to the isolation of the nuclei of the cell line affected by MDS.
Figure 8: Separation of kernels from the pixel.
✓ Step 2: Image Analysis and Processing.
Figure 9 shows the transformations and processing applied to the primary MGm image according to the different dimensions and measurements performed by the CellProfiler software.
Figure 9: Processing of the primary SMD image.
✓ Step 3: Final rendering of cells from the treatment of steps 1 and 2.
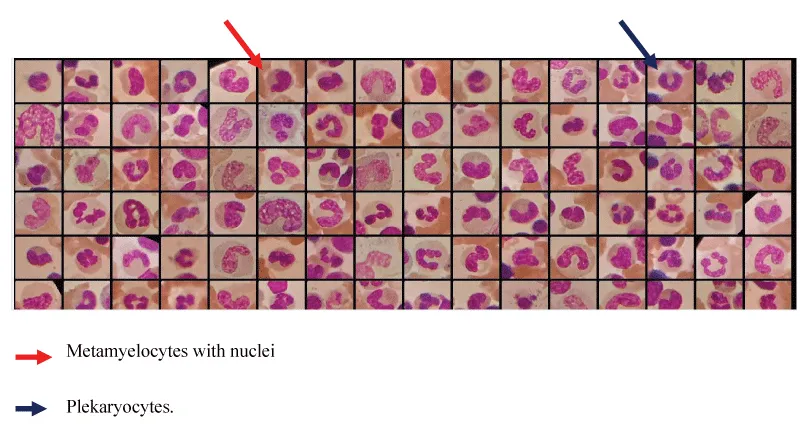
Figure 10 shows the abnormalities of the granulocytic lineage, the most frequently found of which are respectively represented by the red arrow designating metamyelocytes with ribbon or horseshoe nuclei, and the blue arrow designating plekaryocytes [15].
Figure 10: Cellular appearance of bone marrow during MDS.
Vitamin B12 assay
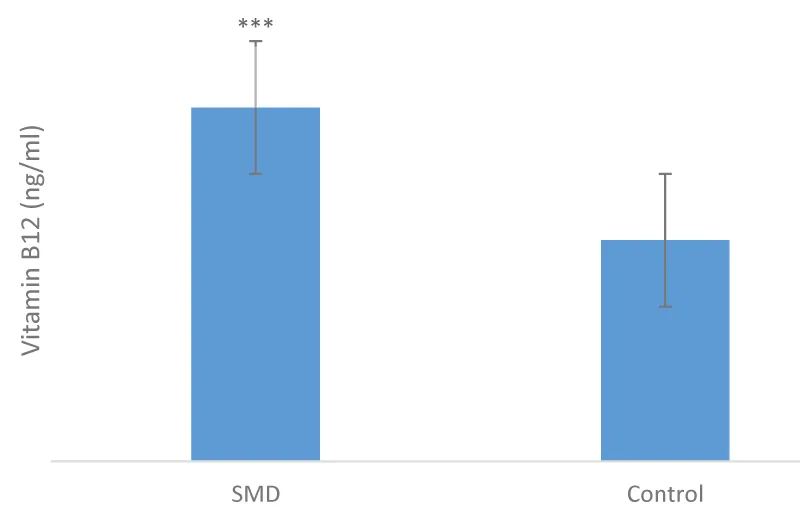
Figure 11 and Table 2 explicitly show the results of the vitamin B12 assay, which showed a non-significant difference between patients and controls; the sample distribution was uneven (p = 0.999).
Figure 11: Distribution of vitamin B12 in patients and controls (p = 0.999), n = 1.
Table 2 illustrates the variation in vitamin B12 levels between patients and controls, marked by an uneven distribution within the sample (p = 0.999) with a mean of 726 ± 612. It also shows the diagnoses of the different cell lines affected in MDS, including the erythrocyte, granulocyte, and thrombocytic cell lines. This table demonstrates that the cell line most affected by MDS in this study was the erythrocyte lineage, the impact of which was observed in the low hemoglobin levels.
Brazzaville, the political capital of the Republic of the Congo, is the largest and most populous city in the country and the only one hosting a hospital specialized in the management of myelodysplastic syndromes (MDS), namely the Clinical Hematology Department of the Brazzaville University Hospital (CHU-B). According to the 2023 census, Brazzaville has an estimated population of approximately 2.1 million inhabitants, representing about 34% - 35% of the national population, which is estimated at 6.1 million. Together with Pointe-Noire, these two major cities account for nearly half of the total population of the Republic of the Congo [16]. Patient recruitment was conducted over five months, from November 2020 to March 2021. The frequency of patients consulting the hematology department of CHU-B for myelodysplastic syndromes (MDS) was 4.59% (8 out of 174 patients). This low proportion highlights the relatively low incidence of MDS in Brazzaville. This result contrasted with that of a study conducted in Algeria in 2014 on MDS in a hospital setting, where a total of 148 patients were identified, including 28 cases of MDS, representing 41.44% [17]. The low frequency of MDS observed in our study could be explained by the very rapid progression of MDS to Acute Myeloid Leukemia (AML), which is one of the complications of MDS when it is diagnosed late.
The mean age of the study population (Table 2) was 51.2 ± 12.3 years, with a range of 30 years. This finding contrasts with data from Europe in 2008, where the most affected age group was over 70 years [2]. Our results suggest that MDS is not solely a disease of the elderly, but can affect a broader age range.
Regarding sex distribution (Table 2), males predominated, accounting for 62.5% of the population, while females represented 37.5% (p = 1.000). This aligns with European data from 2013, which reported that MDS preferentially affects men, with a sex ratio of 1.8 [2]. However, the underlying reason for this male predominance remains unclear, representing a limitation of the study. Given the very low number of positive cases (8) included in this study, primarily due to the rarity of the disease, despite a five-month study period, this work remains a preliminary study that encourages a more comprehensive future investigation with a larger sample size.
The most common reasons for consultation among the study population were asthenia, dyspnea, and persistent fevers. This finding is similar to that observed in France in 2005, which concluded that myelodysplastic syndromes (MDS) cause dysfunction in one or all blood cell lines [7]. This could respectively manifest as damage to the erythrocyte lineage, potentially leading to asthenia, decreased hemoglobin levels, and dyspnea due to oxygen deficiency; and damage to the granulocytic lineage, specifically neutrophils (PMNs), which are the body’s primary defense agents, consequently causing persistent fevers [17].
The morphological appearance of blood cells in MDS was described quantitatively using the complete blood count (CBC) and qualitatively using peripheral blood smears, bone marrow aspirates, and exclusive cell pooling of bone marrow.
The CBC results accurately showed the variation in hemoglobin levels, leukocyte counts, and platelet counts in our study sample.
The hemoglobin levels of the patients were very low compared to those of the controls; a highly significant difference was observed (p < 0.001). The mean Hb level in the cases was 7.47 ± 1.42 g/dL. This value was approximately equivalent to that found in Algeria in 2014, which was 8.34 ± 2 g/dL [17]. This low hemoglobin level could explain the involvement of the erythrocyte lineage responsible for the recurrent anemia observed in myelodysplastic syndromes (MDS).
Indeed, regarding the patients’ leukocyte count, the mean was approximately 3.19 ± 1.89, compared to 4.18 ± 0.75 in the controls. There was no significant difference between cases and controls. This could indicate that in MDS, the leukocyte count alone is insufficient to assess infection; it must be compared to the quality of the leukocytes as revealed by the bone marrow aspirate.
Furthermore, regarding the platelet count, according to the Student’s t-test, the complete blood count results showed no significant difference between the platelet counts of patients and controls (p = 0.428). This result could suggest that in MDS, involvement of the thrombocytic lineage is optional and can be associated with the observed anemia. As stated in France in 2004, myelodysplastic syndromes (MDS) can affect either the white blood cell lineage, the red blood cell lineage, or the platelet lineage; however, they can also affect all three lineages simultaneously [18].
The results concerning blood cell quality in MDS were more clearly elucidated by bone marrow aspirate and selective pooling of pathological blood cells from the bone marrow (BM) using Cell Profiler than by peripheral blood smear. This BM cell quality analysis showed not only the appearance and presence of metamyelocytes with ribbon-like nuclei, but especially the presence of plekaryocytes, which are, among other things, responsible for the dysfunction observed within neutrophils (PMNs). This could explain the cause of the recurrent infections resulting in persistent fevers during MDS [17].
The results of the vitamin B12 assay showed no significant difference between groups, reflecting an uneven distribution within the sample (p = 0.999). The variation in vitamin B12 levels between patients and controls was comparable, suggesting that neither excess nor deficiency of vitamin B12 is consistently associated with the presence of MDS. However, it is noteworthy that two cases within the study population exhibited markedly elevated vitamin B12 levels (2000 pg/ml), substantially exceeding the normal reference range of 200–500 pg/ml.
Future studies should further investigate the potential role of additional factors, including deficiencies in folate (vitamin B9) and vitamin B6, as well as metabolic disorders such as hyperhomocysteinemia, which may contribute to the pathogenesis of myelodysplastic syndrome (MDS) through impaired hematopoiesis [19]. Moreover, factors affecting nutrient absorption—such as autoimmune disorders, chronic alcohol consumption, or medications like metformin—that could exacerbate megaloblastic anemia should also be taken into consideration [20-22].
In conclusion, it is necessary to state that myelodysplastic syndromes (MDS) are silently prevalent in our country, with a hospital incidence of 4.59% in Brazzaville. The morphology of blood cells in this pathology reveals the presence of metamyelocytes with ribbon-like nuclei and plekaryocytes. The excess or deficiency of vitamin B12 observed in MDS is not responsible for its unexpected onset; therefore, it is important to conclude that vitamin B12 has no close relationship with the development of MDS.
- Afsaneh B, Mikael A. Myelodysplastic syndromes: a practical approach to diagnosis and treatment. Cleve Clin J Med. 2010;77(1). Available from: https://doi.org/10.3949/ccjm.77a.09069
- Germing U, Kobbe G, Haas R, Gattermann N. Myelodysplastic syndromes: diagnosis, prognosis, and treatment. Dtsch Arztebl Int. 2013;110(46):783–790. Available from: https://doi.org/10.3238/arztebl.2013.0783
- Maynadié M, Girodon F, Manivet-Janoray I, Mounier M, Mugneret F, Bailly F, et al. Twenty-five years of epidemiological recording on myeloid malignancies: data from the specialized registry of hematologic malignancies of Côte d’Or (Burgundy, France). Haematologica. 2011;96(1):55–61. Available from: https://doi.org/10.3324/haematol.2010.026252
- Rollison DE, Howlader N, Smith MT, Strom SS, Merritt WD, Ries LA, et al. Epidemiology of myelodysplastic syndromes and chronic myeloproliferative disorders in the United States, 2001–2004, using data from the NAACCR and SEER programs. Blood. 2008;112(1):45–52. Available from: https://doi.org/10.1182/blood-2008-01-134858
- Arber DA, Orazi A, Hasserjian R, Thiele J, Borowitz MJ, Le Beau MM, et al. The 2016 revision to the World Health Organization classification of myeloid neoplasms and acute leukemia. Blood. 2016;127(20):2391–2405. Available from: https://doi.org/10.1182/blood-2016-03-643544
- Belakehal S, Bekadja S, Touhami H, Hamladji RM, Mesli N. Epidemiological approach to myelodysplastic syndromes in Algeria (1995–2005): a cooperative and multicenter study. 2006. SAHTs.
- Provan D, Gribben JG. Molecular hematology. 2nd ed. Blackwell Publishing; 2005;268.
- Bryon PA. Anatomy and histology of the bone marrow. EMC Hematology. 1998;10. Article 13-92000-M-80.
- Sebahaum G, Troussard X. Cytology and histology of the bone marrow. Hematology. Elsevier Masson SAS; 2010. Article 13-000-a-30.
- Turhan AG, Humphries RK, Phillips GL. Clonal hematopoiesis demonstrated by X-linked DNA polymorphisms after allogeneic bone marrow transplantation. N Engl J Med. 1989;320:1655–1661. Available from: https://doi.org/10.1056/nejm198906223202504
- Hillman RS, Ault KA, Rinder K. Hematology in clinical practice: a guide to diagnosis and treatment. French ed. Flammarion SA; 2007. Available from: https://books.google.co.in/books?id=yfFLAQAAIAAJ&lr=&num=20&source=gbs_book_other_versions_r&cad=3
- Hamri L. Myelodysplastic syndromes (about 21 cases). 2015;35–40.
- Tefferi A, Vardiman JW. Myelodysplastic syndromes. N Engl J Med. 2009;361(19):1872–1885. Available from: https://doi.org/10.1056/nejmra0902908
- Ross MH, Wilson KJ. Anatomy and physiology in health and illness. 11th ed. Elsevier Masson; 2019;60–68. Available from: https://pharmabookbank.wordpress.com/wp-content/uploads/2019/03/1.2.ross-wilson-anatomy-and-physiology.pdf
- Lessard M, Gervais C, Struski S. Cytogenetic abnormalities of myelodysplastic syndromes and secondary acute leukemias. Pathol Biol (Paris). 2003;51(6):356–365.
- National Institute of Statistics. Preliminary results of the 5th General Population and Housing Census. RGPH-5 report. December 2023.
- Brechemier D. Impact of comorbidities on the prognosis of patients over 75 years of age treated with azacitidine for high-risk myelodysplastic syndrome or acute myeloblastic leukemia. June 2014;24.
- Dewulf G, Gouin I, Pautas E, Gaussem P, Chaïbi P, Andreux JP, et al. Myelodysplastic syndromes diagnosed in a geriatric hospital: cytological profile of 100 patients. Ann Biol Clin (Paris). 2004;62(2):197–202. Available from: https://pubmed.ncbi.nlm.nih.gov/15047472/
- Feger F, Vainchenker W. Hematopoiesis and growth factors. EMC Hematology. 1997:1–10. Article 13-000-M-85.
- Lorand-Metze I, Ribeiro E, Lima CSP, Suárez L, Batista K, Metze K. Detection of hematopoietic maturation abnormalities by flow cytometry in myelodysplastic syndromes and its utility for the differential diagnosis with non-clonal disorders. Leuk Res. 2007;31:147–155. Available from: https://doi.org/10.1016/j.leukres.2006.04.010
- Kaushansky K, Lichtman MA, Beutler E, Kipps TJ, Seligsohn U, Prchal JT. Williams hematology. 8th ed. McGraw-Hill Companies; 2010;1523.
- Najman A, Verdy E, Potron G, Isnard F. Handbook of blood diseases. Vol. I. Marketing Edition; 1994.