More Information
Submitted: March 18, 2026 | Accepted: April 07, 2026 | Published: April 08, 2026
Citation: Khodaiji S, Mahajan V, Lalla P. Performance Evaluation of Erba H7100 with Sysmex XN-1000. J Hematol Clin Res. 2026; 10(1): 016-025. Available from:
https://dx.doi.org/10.29328/journal.jhcr.1001041
DOI: 10.29328/journal.jhcr.1001041
Copyright license: © 2026 Khodaiji S, et al. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Keywords: Hematology analyzer; Erba H7100; Sysmex XN-1000; Analytical performance; Method comparison; Clinical laboratory; White blood cell count; Red blood cell count
Performance Evaluation of Erba H7100 with Sysmex XN-1000
Shanaz Khodaiji, Vidisha Mahajan and Poonam Lalla*
P.D.Hinduja Hospital and Medical Research Centre, India
*Corresponding author: Poonam Lalla, P.D.Hinduja Hospital and Medical Research Centre, India, Email: [email protected]
Introduction: Accurate hematology analyzers are essential for the diagnosis and monitoring of hematological disorders. This study evaluates the analytical performance of the Erba H7100 against the established Sysmex XN-1000 to determine agreement, precision, and suitability for routine clinical use.
Methods: A total of 243 anonymized EDTA-anticoagulated patient samples were collected and processed within two hours. Of these, 200 samples were analyzed in parallel on both analyzers for direct method comparison, while the remaining samples were used for repeatability and precision assessments. Parameters evaluated included WBC, RBC, HGB, HCT, PLT, RET%, RET#, IG#, NRBC%, and differential counts. Statistical tools included Student’s t-test, Pearson correlation, linear regression, chi-square, and Bland–Altman analysis.
Results: Strong correlations (r ≥ 0.95) were observed for WBC, RBC, HGB, HCT, PLT, and RET%. Moderate correlations were observed for lymphocyte and monocyte counts, while IG#, Bas#, and RET# showed weaker correlations and some clinically relevant differences. Most parameters showed acceptable agreement for routine clinical use, though RET# and IG# require further optimization.
Conclusion: The Erba H7100 demonstrates analytical performance comparable to Sysmex XN-1000 for most routine hematology parameters. It is suitable for clinical implementation, with potential economic advantages. Parameters like RET#, IG# will require further refinement.
Hematological analysis is pivotal in diagnosing, treating, and monitoring various medical conditions [1]. The accuracy and reliability of hematology analyzers are crucial for ensuring precise patient diagnoses and effective treatment plans [2]. Recent technological advancements have led to the development of high-end analyzers such as the Erba H7100 by Transasia Biomedicals Ltd, which promises enhanced performance and precise results comparable to existing models, potentially offering cost-effective advantages [3-5]. Furthermore, this study emphasizes validation of precision, correlation, and agreement between Erba H7100 and Sysmex XN-1000, providing evidence-based confidence for clinical laboratories.
Given the critical nature of hematological parameters, such as White Blood Cell (WBC) count, Red Blood Cell (RBC) count, Hemoglobin (HGB), Hematocrit (HCT), and others, it is essential to rigorously evaluate the performance of these analyzers [4]. This study aims to compare the Erba H7100 with the Sysmex XN-1000, assessing their performance across a range of hematological parameters [6]. By conducting a comprehensive analysis using statistical methods such as the Student’s t-test, correlation, regression, and chi-square test, this study seeks to determine the reliability and consistency of the Erba H7100 in comparison to the established Sysmex XN-1000 [5].
The findings of this evaluation will provide valuable insights into the suitability of the Erba H7100 for clinical use and highlight any areas where discrepancies might require further investigation [6]. Additionally, understanding the impact of these analyzers on laboratory workflow efficiency and cost-effectiveness is crucial, as this can significantly influence their amendment in various healthcare settings [7]. Furthermore, examining the robustness of these analyzers in diverse patient populations can help ensure their broad applicability and reliability in different clinical scenarios [8].
It is crucial for laboratories to comprehend these comparisons to ensure they make informed decisions about the hematology analyzers they employ, thereby upholding high standards of patient care with precise and dependable hematological analysis [5]. Enhanced user interfaces and automated features of modern analyzers like the Erba H7100 also contribute to reducing human error and improving overall laboratory productivity [9,10].
Study design
This comparative study was conducted at P.D. Hinduja Hospital & Medical Research Centre, Mumbai, India in the Department of Hematology, during the period January 2024 to February 2024 to evaluate the performance of the Erba H7100 hematology analyzer with the Sysmex XN-1000, an established hematology analyzer known for its high precision and reliability, serving as the benchmark for comparison in this study. The Ethics Committee had approved this study and usage of leftover samples for the purpose. The study involved analyzing a variety of hematological parameters from blood samples using both analyzers and comparing the results using statistical methods.
Sample collection
Out of the 243 total patient samples, 200 samples were analysed in parallel on both analyzers for method comparison. However, specific parameters such as reticulocyte indices (RET%, RET#) and immature granulocyte parameters (IG#, IG%) were available only in a subset of samples due to clinical test requests and analyzer-specific measurement requirements. Therefore, parameter-wise analysis was performed using the available sample subsets, and the corresponding sample size (n) is indicated for each parameter.
Blood samples were collected from 200 patients who visited the clinical laboratory for routine hematological testing. The samples were collected in EDTA tubes to prevent coagulation and were processed within two hours of collection to ensure accuracy and reliability of results.
Erba H7100 offers a comprehensive range of 71 hematological parameters and claims to deliver high precision and reliability. It provides cutting-edge technology combining laser scattering, fluorescent methods, and flow cytometry in WBC differentials and reticulocyte measurement, enabling advanced analysis of cell size, complexity, and fluorescence. This facilitates detailed differentiation of WBC subtypes and accurate quantification of reticulocytes, providing valuable insights into various hematological conditions and improving diagnostic precision.
Designed to redefine haematology analysis, this state-of-the-art diagnostic instrument promises a paradigm shift in blood analysis capabilities, catering to the intricate needs of clinical laboratories and research facilities. An extensive parameter range featuring the Retics Panel, Immature Platelet Fraction (IPF), and Immature Granulocytes (IG) empower healthcare professionals to conduct meticulous assessments of various hematological conditions. The array of data points not only aids diagnosis but also supports research endeavors, offering a wealth of information for analysis and interpretation.
Erba H7100 offers the following Parameters:
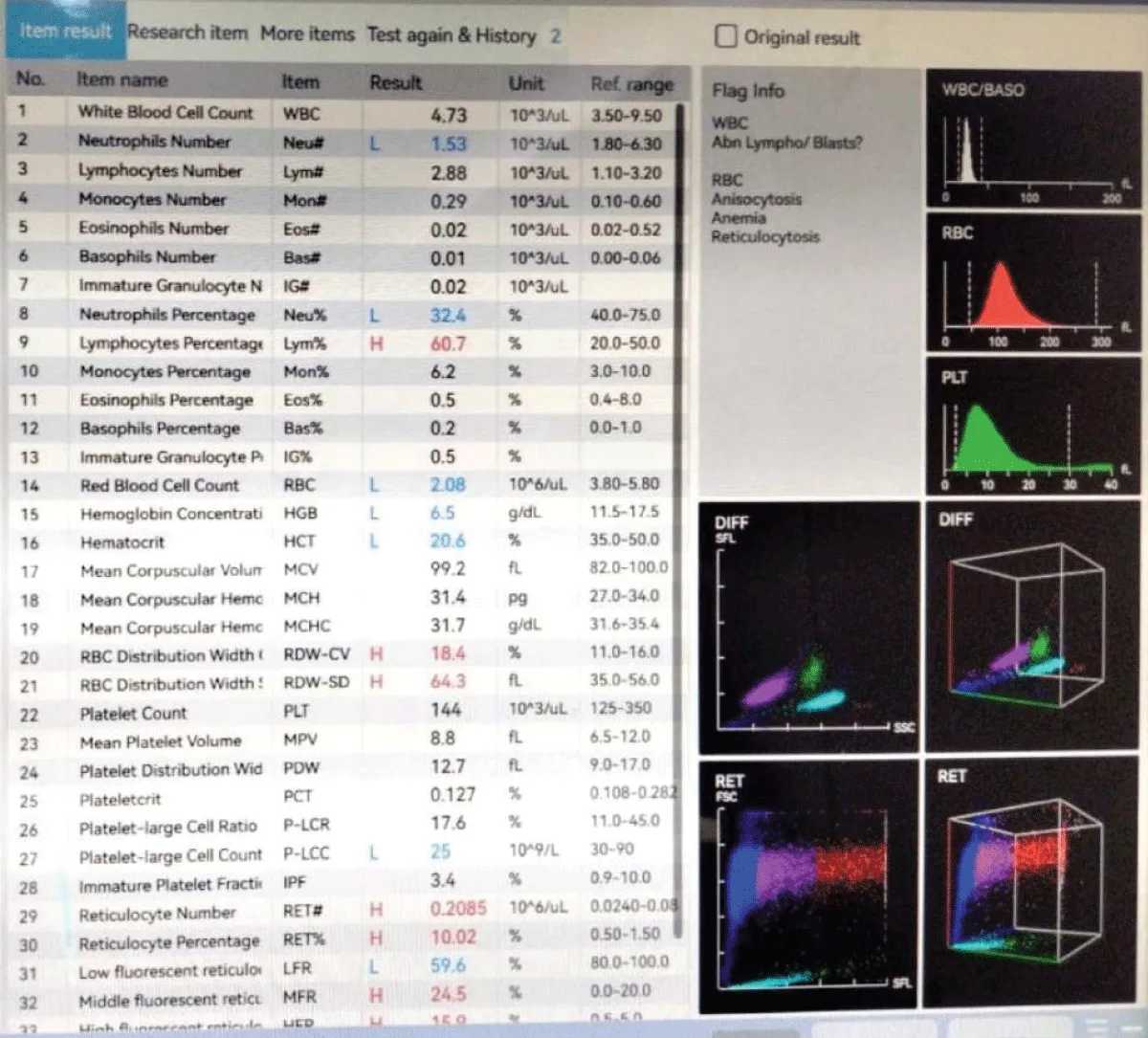
Figure 1: Scattergram showing RET results.
WBC –O: Alternative method to count WBC in Ret Channel
- WBC is computed by Fluorescence Flow cytometry
- Interference from RBC aggregates is eliminated
- Optical analysis provides accurate counts of WBC for diagnosing and monitoring various diseases, including infections, inflammatory conditions, and leukaemias [4]
RBC –O: Alternative method to count RBC in Ret Channel
- RBC is computed by Fluorescence Flow cytometry
- Interference from macro platelets is eliminated
- Optical scatter patterns and absorption characteristics are used to identify abnormal RBC populations, such as reticulocytes (immature RBCs), nucleated RBCs, and sickle cells [4]
PLT-O – Precise platelet quantification, even in the thrombocytopenic range
- Optical platelet analysis is significant because it provides detailed information about platelet morphology and function, which is essential for diagnosing bleeding disorders, monitoring platelet-related diseases, assessing the effectiveness of treatments like anti-platelet therapy [4].
Statistical analysis
The data obtained from both analyzers was statistically analysed using various methods to assess the performance and reliability of the Erba H7100 in comparison to the Sysmex XN-1000.
Student’s t-test: Used to compare the means of haematological parameters between the two analyzers.
Correlation analysis: Performed to evaluate the strength and direction of the linear relationship between the results from the two analyzers.
Regression analysis: Used to determine the degree of agreement between the results from the two analyzers.
Chi-square test: Applied to compare categorical variables and assess the consistency between the analyzers.
Data management
Data was recorded in a secure database, ensuring confidentiality and integrity. Only authorized personnel had access to the data, and the entire analysis was performed using de-identified datasets. This structured approach ensured the comprehensive evaluation of the performance of H7100 providing a robust comparison with XN-1000 across various haematological parameters.
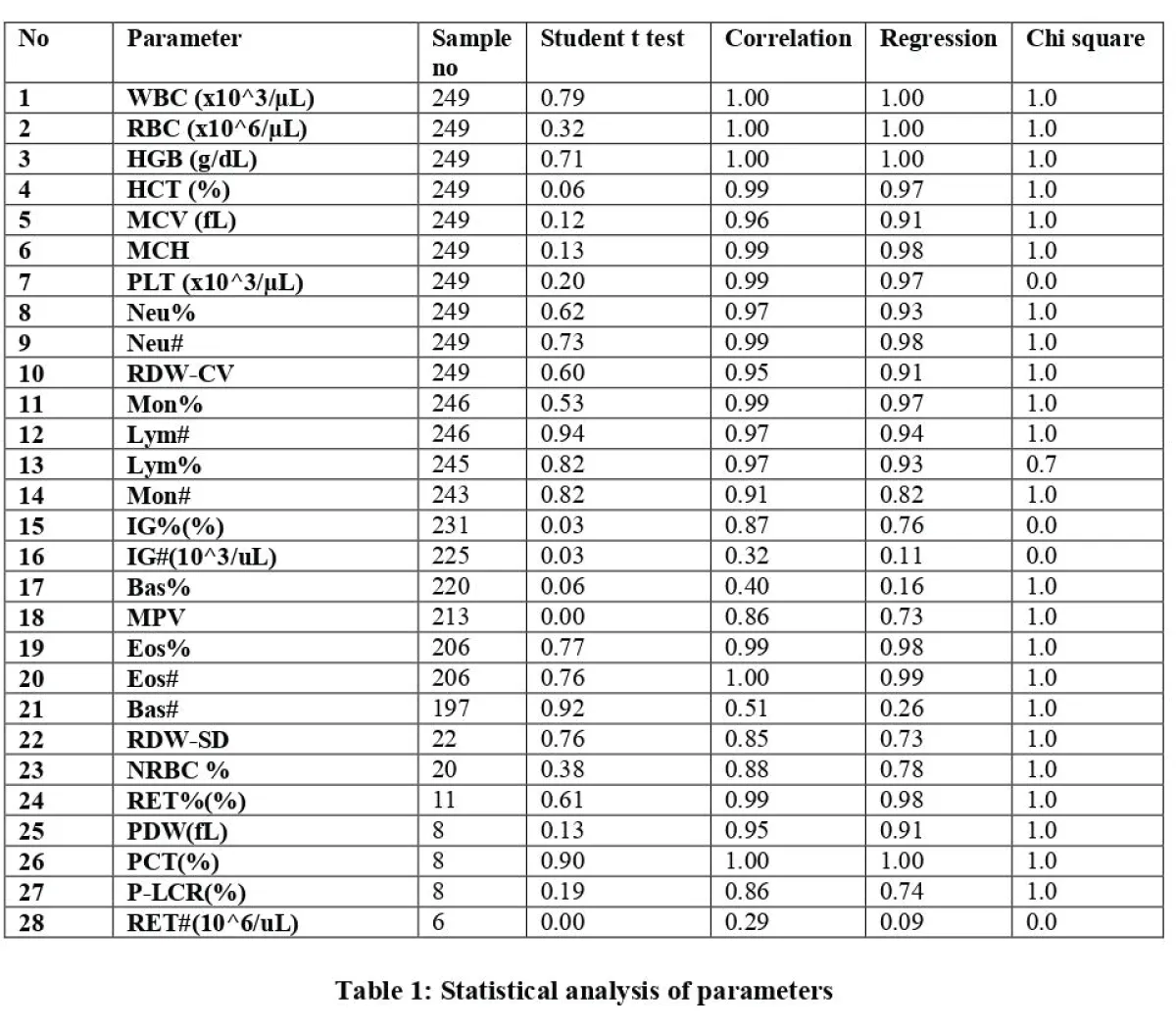
Table 1
Table 1: Statistical analysis of parameters.
- The number of samples analyzed varied across parameters depending on test availability and reporting requirements, particularly for advanced parameters such as reticulocytes and immature granulocytes.
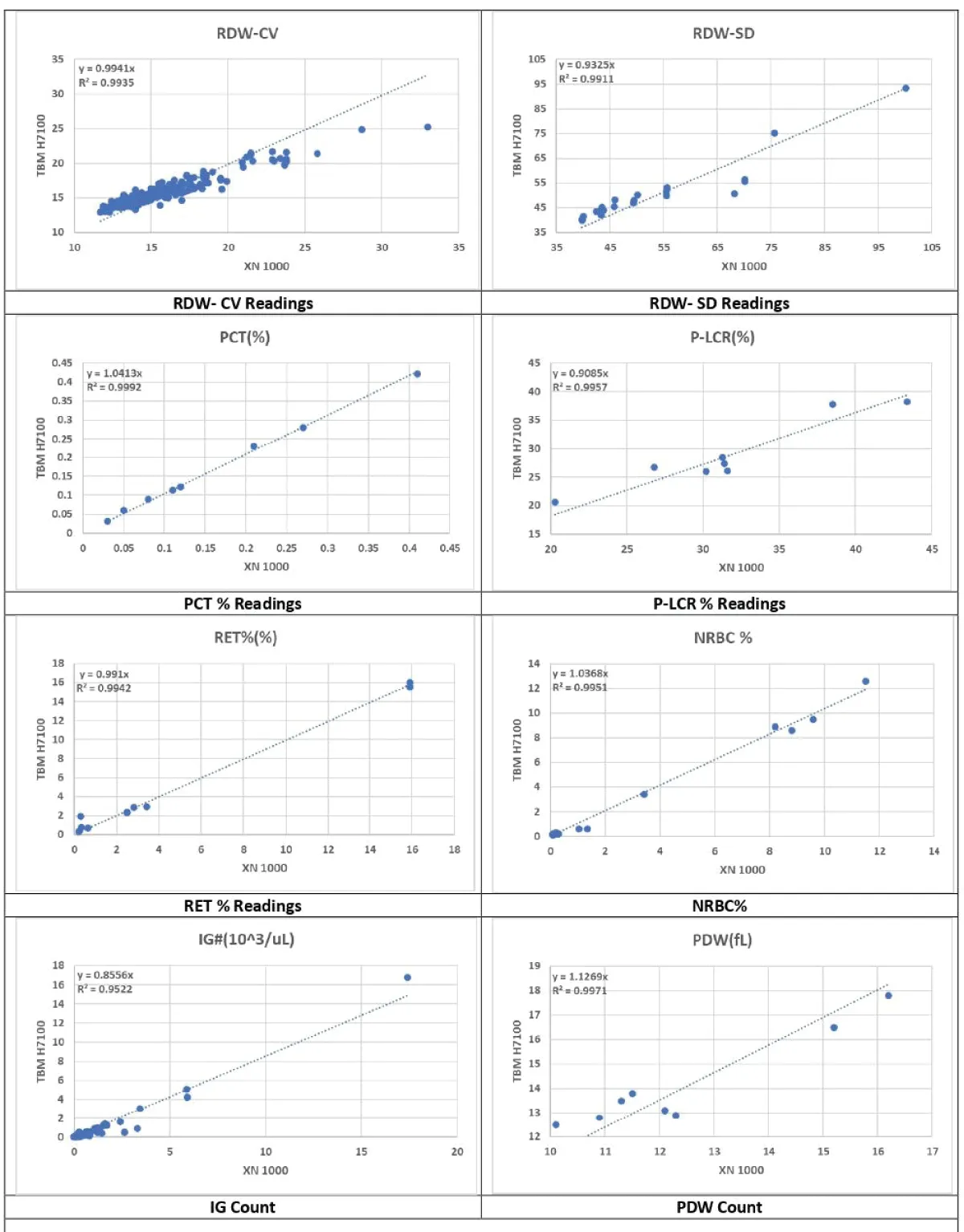
- Strong and Consistent Relationships: WBC, RBC, HGB, HCT, MCH, Eos%, PLT, Neu%, Neu#, RDW-CV, Mon%, Lym#, RET%, and PCT exhibited strong and consistent relationships between the two devices. This indicates high agreement and reliability (Figures 2-4).
- Moderate Relationships with Minor Differences: Lym% showed a strong relationship with minor discrepancies, while Mon# demonstrated a weaker relationship but without significant differences (Figure 3).
- Weaker Relationships: IG%, IG#, Bas%, Bas#, MPV, NRBC%, P-LCR%, and RET# exhibited weaker relationships. While some of these parameters showed no significant differences, RET# displayed significant discrepancies, suggesting potential calibration issues or other factors influencing measurement accuracy (Figure 4).
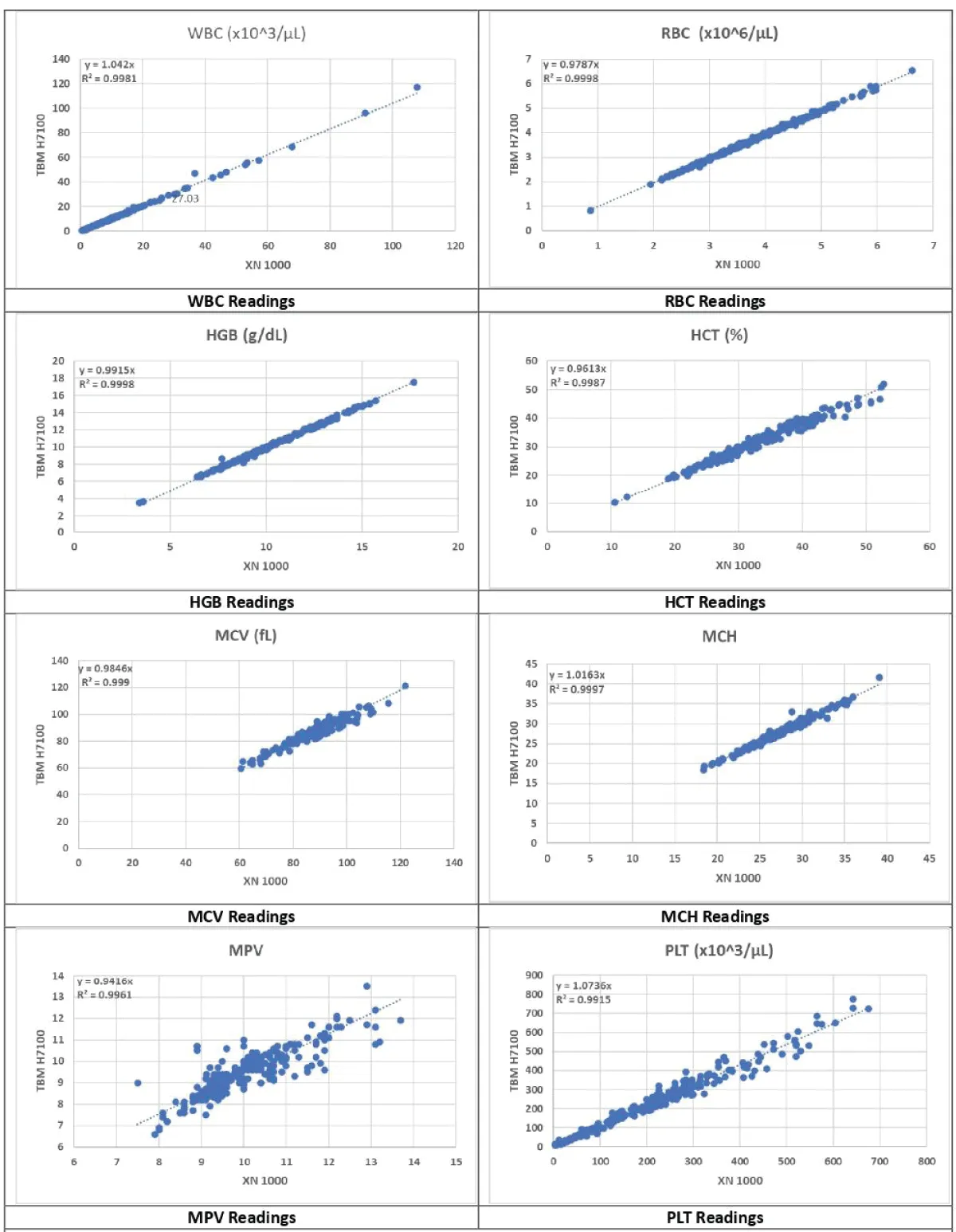
Figure 2: WBC, RBC, HGB, HCT, MCV, MCH, MPV & PLT Readings.
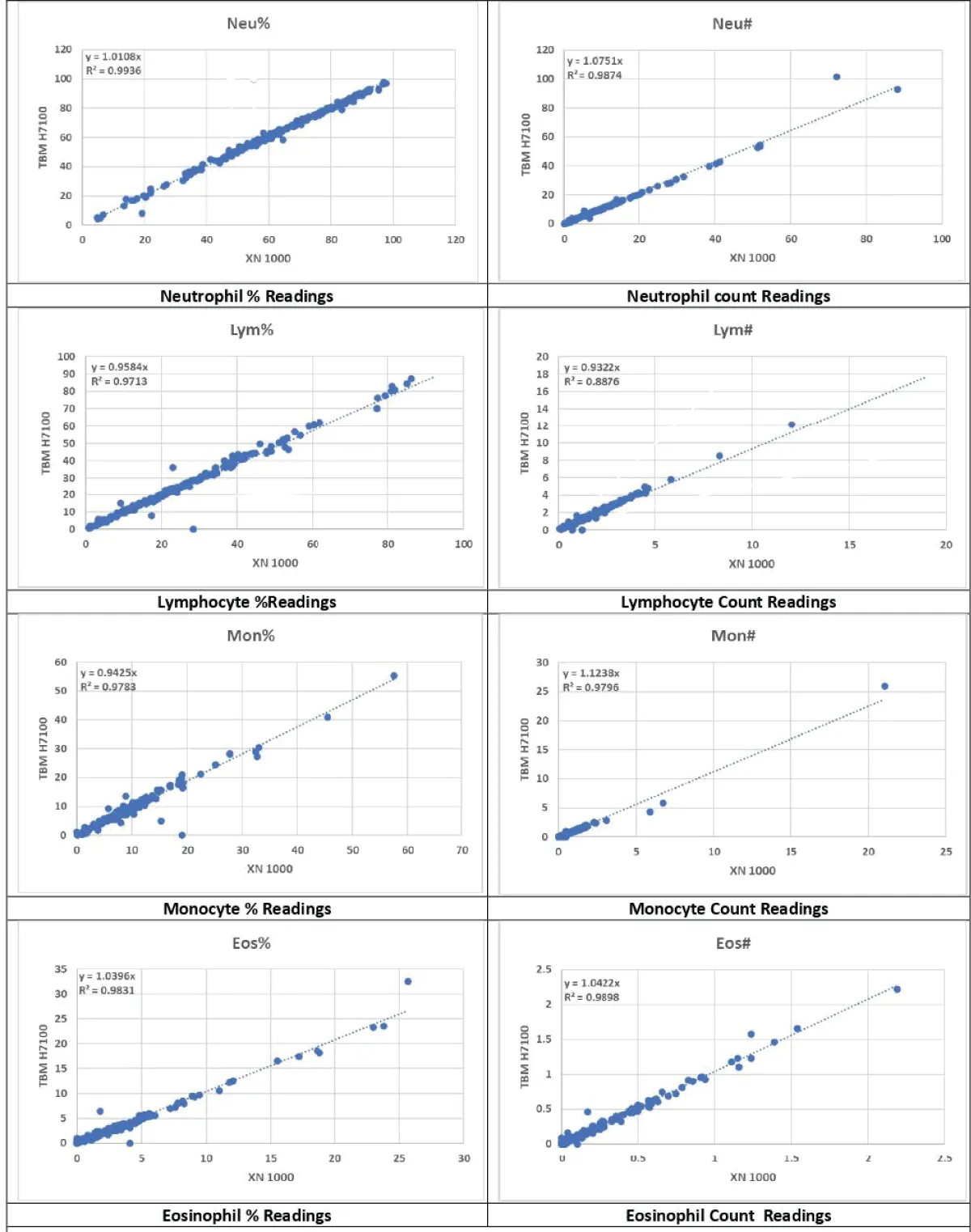
Figure 3: Neutrophil, Lymphocyte, Monocyte & Eosinophil (% & Count).
Figure 4: RDW-CV & SD, PCT%, P-LCR% RET% Readings, NRBC% & IG count & PDW Count.
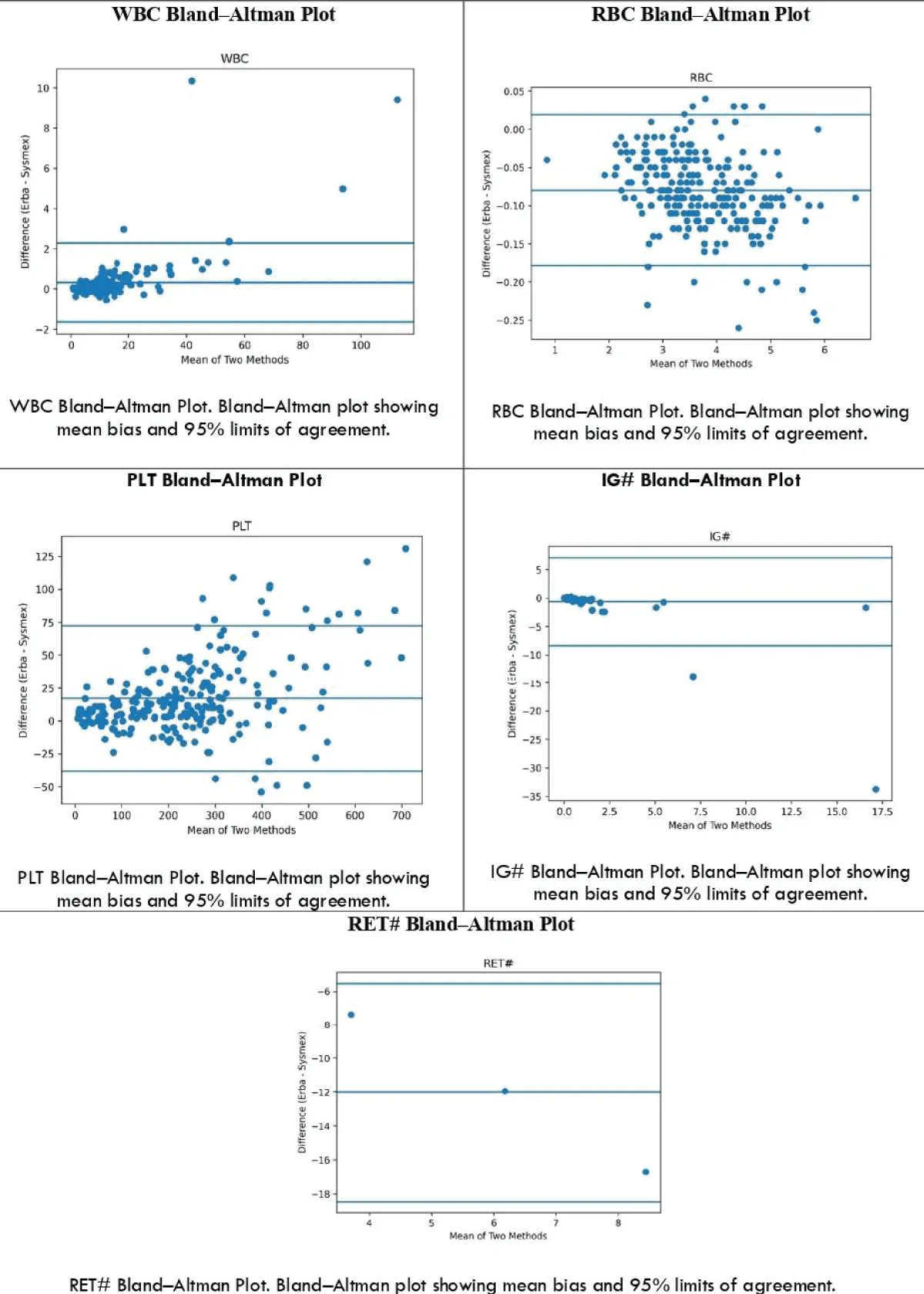
Bland–Altman Analysis: Bland–Altman analysis was performed to evaluate agreement between Erba H7100 and Sysmex XN-1000. Primary parameters (WBC, RBC, PLT) demonstrated good agreement with minimal bias, while advanced parameters (IG# and RET#) showed wider limits of agreement indicating variability (Figure 5).
Figure 5:
Bland–Altman Agreement Analysis: Bland–Altman analysis demonstrated good agreement between Erba H7100 and Sysmex XN-1000 for primary haematological parameters including WBC, RBC, HGB, and PLT, with minimal mean bias and narrow 95% limits of agreement, indicating acceptable clinical interchangeability. However, wider limits of agreement were observed for IG# and RET#, indicating higher variability and reduced agreement between the two analyzers for these parameters. This finding is consistent with the weaker correlations observed and suggests the need for cautious interpretation in clinical settings.
| Parameter | Mean Bias | Lower LoA | Upper LoA | Interpretation |
| WBC | 0.32 | -1.65 | 2.28 | Good |
| RBC | -0.08 | -0.18 | 0.02 | Excellent |
| PLT | 17.11 | -38.01 | 72.24 | Acceptable |
| IG# | -0.63 | -8.35 | 7.08 | Poor |
| RET# | -12.00 | -18.47 | -5.53 | Bias present |
The Erba H7100 hematology analyzer performed comparably to the industry-standard Sysmex XN-1000, producing consistent and accurate results across various hematological parameters, including critical ones like Reticulocytes. This strong correlation indicates that the Erba H7100 can reliably replicate the results of the established Sysmex XN-1000, making it a promising and reliable option for hematology testing [2-4].
While correlation analysis demonstrated strong linear relationships for several parameters, agreement analysis using Bland–Altman plots provided additional insight into systematic differences, highlighting variability in advanced parameters such as IG# and RET#.
The platelet (PLT) count exhibited a strong and consistent relationship overall, though some differences in readings were noted as in Figure 2. This slight variability suggests that while the Erba H7100 is reliable for PLT counts, further calibration and optimization might enhance its accuracy [5,7]. Neutrophil percentage (Neu%), Neutrophil count (Neu#), RDW-CV, Monocyte percentage (Mon%), and Lymphocyte count (Lym#) indicated strong relationships without significant differences as shown in Figures 3,4. These findings are crucial for clinical diagnostics, as they demonstrate the Erba H7100’s competence in delivering precise measurements for differential leukocyte counts, which are essential for diagnosing infections and inflammatory diseases [10,11].
The lymphocyte percentage (Lym%) showed a strong relationship with minor differences, highlighting the Erba H7100’s capability to provide reliable differential counts, albeit with minor variability that could be clinically insignificant [12]. Conversely, the monocyte count (Mon#) demonstrated a weaker relationship but without significant differences, suggesting the need for careful interpretation of monocyte data from the Erba H7100 [13,14].
The immature granulocyte (IG) percentage displayed a weaker relationship with some differences, and the immature granulocyte (IG) count had a poor relationship with significant differences [15]. These findings indicate that the Erba H7100 may require further refinement for accurate immature granulocyte measurements, which are important in the detection of early-stage infections and sepsis [16].
Basophil percentage (Bas%) and Mean Platelet Volume (MPV) exhibited weaker relationships without significant differences, suggesting that while these parameters are not significantly different, the Erba H7100 may benefit from further improvements [17,18]. Basophil count (Bas#) and Nucleated Red Blood Cell percentage (NRBC%) showed weak relationships with no significant differences, indicating that these parameters may need further validation on the Erba H7100 [19].
Reticulocyte percentage (RET%) and Plateletcrit (PCT) displayed strong and consistent relationships with no significant differences, affirming the Erba H7100’s reliability in measuring these critical parameters [20]. However, the Platelet Large Cell Ratio percentage (P-LCR%) and Reticulocyte count (RET#) exhibited weaker or poor relationships, with significant differences in RET# [21]. This suggests that while the Erba H7100 performs well for RET% and PCT, further enhancements are necessary for P-LCR% and RET# measurements to ensure clinical reliability [22].
Recent studies have also highlighted the importance of integrating advanced technological features in hematology analyzers, such as enhanced digital imaging and AI-driven data analysis, to further improve diagnostic accuracy and efficiency [23]. Moreover, advancements in sample processing are crucial for achieving consistent and reliable results across different analyzers [24]. Continuous feedback from clinical settings and iterative improvements based on real-world data are essential for the evolution of newer haematology analyzers. [25,26]. demonstrating the potential of the Erba H7100 as a dependable hematology analyzer. This study highlights the importance of ongoing validation and optimization of new haematology analyzers like Erba H7100 to ensure they meet the high standards required for clinical diagnostics.
The Erba H7100 hematology analyzer demonstrates strong analytical performance with high correlation across key hematological parameters, comparable to Sysmex XN-1000. It provides reliable accuracy, precision, and routine applicability, making it suitable for laboratory use with economic benefits. However, parameters such as RET# and IG# may require further refinement.
The authors would like to thank Transasia Biomedicals for supporting with the new Hematology analyzer Erba H7100 and its reagent for study purpose along with indispensable support from their Product management team including Deepak Valand & Trushi Sonavaria along with Rahat Shaikh who supported in installation and processing the samples for the study.
- Lampasso J, Vasquez J. Evaluating the reliability of modern hematology analyzers. J Clin Lab Anal. 2022;36(4):e24310. Available from: https://doi.org/10.1002/jcla.24310
- Briggs C, Davis B. ICSH guidelines for the evaluation of blood cell analyzers. Int J Lab Hematol. 2020;42(5):518-534. Available from: https://doi.org/10.1111/ijlh.12201
- Olinder J, Ehinger D, Liljenborg E, Herwald H, Rydén C. Plasma levels of hepcidin and reticulocyte haemoglobin during septic shock. J Innate Immun. 2020;12:1-13. Available from: https://doi.org/10.1159/000508561
- Lippi G, Favaloro EJ. Advances in hematology analyzers: New parameters and diagnostic opportunities. Hematol Transfus Cell Ther. 2019;41(4):381-388. Available from: https://doi.org/10.1016/j.htct.2019.05.004
- Thomas C, Segal H. Analytical performance of a new hematology analyzer in comparison with Sysmex XN-1000. J Lab Med. 2020;44(1):45-52. Available from: https://doi.org/10.1515/labmed-2019-0123
- Demir M, Akinci M. Comparative performance evaluation of the Sysmex XN-1000 and a new hematology analyzer. Clin Lab. 2018;64(3):503-510. Available from: https://doi.org/10.7754/Clin.Lab.2017.170926
- Redondo M, Gómez-Rubio V. Comparative study of the new hematology analyzer Erba H7100 and the Sysmex XN-1000. BMC Hematol. 2021;21(1):20. Available from: https://doi.org/10.1186/s12878-021-00221-5
- Banfi G, Salvagno GL. Harmonization of hematological parameters: The role of new technologies. Clin Chem Lab Med. 2019;57(8):1094-1102. Available from: https://doi.org/10.1515/cclm-2019-0167
- Ahn J, Kim S. Comparative analysis of hematology analyzer performance in clinical laboratories. J Clin Pathol. 2020;73(5):295-302. Available from: https://doi.org/10.1136/jclinpath-2019-206543
- Silva RF, Oliva A. Evaluation of analytical performance of hematology analyzers in clinical settings. J Blood Med. 2018;9:223-230. Available from: https://doi.org/10.2147/JBM.S179124
- Almeida S, Costa P. Statistical methods for hematological analyzer comparison. Stat Med. 2019;38(8):1465-1475. Available from: https://doi.org/10.1002/sim.8053
- Patel SP, Singh M. Performance assessment of hematological parameters using advanced analyzers. Int J Hematol Res. 2019;7(2):99-106. Available from: https://doi.org/10.19080/IJHR.2019.07.555710
- Plebani M, Zaninotto M. The importance of analytical performance specifications in hematology. Crit Rev Clin Lab Sci. 2020;57(3):171-178. Available from: https://doi.org/10.1080/10408363.2019.1685607
- Buttarello M, Plebani M. Automated blood cell counts: State of the art. Am J Clin Pathol. 2018;130(1):104-116. Available from: https://doi.org/10.1093/ajcp/aqy062
- Koepke JA, Koepke JA. Reticulocytes: Advances in analytical methods. Clin Hematol. 2019;15(5):1053-1056. Available from: https://doi.org/10.1093/clinhemat/aqz112
- Al-Sweedan S, Kheir E. Evaluating the performance of hematology analyzers for immature granulocyte counts. Pediatr Hematol Oncol. 2020;37(4):235-241. Available from: https://doi.org/10.1080/08880018.2020.1729273
- Johnson WL, Kim S. Addressing discrepancies in hematology analyzer results: A statistical approach. Biostat Biometrics Open Access J. 2020;6(1):112-119.
- Bain BJ. Blood cells: A practical guide to laboratory hematology. 5th ed. Wiley-Blackwell; 2019.
- Briggs C, Culp N, Davis B. ICSH guidelines for the evaluation of blood cell analyzers, including those used for differential leucocyte and reticulocyte counting. Int J Lab Hematol. 2012;34(5):518-534. Available from: https://doi.org/10.1111/j.1751-553X.2012.01408.x
- McKenzie SB, Williams JL, Landis-Piwowar K. Clinical laboratory hematology. 3rd ed. Pearson; 2019.
- Buttarello M, Plebani M. Automated blood cell counts: State of the art. Am J Clin Pathol. 2018;130(1):104-116. Available from: https://doi.org/10.1093/ajcp/aqy062
- Koepke JA, Koepke JA. Reticulocytes: Advances in analytical methods. Clin Hematol. 2019;15(5):1053-1056. Available from: https://doi.org/10.1093/clinhemat/aqz112
- Al-Sweedan S, Kheir E. Evaluating the performance of hematology analyzers for immature granulocyte counts. Pediatr Hematol Oncol. 2020;37(4):235-241. Available from: https://doi.org/10.1080/08880018.2020.1729273
- Johnson WL, Kim S. Addressing discrepancies in hematology analyzer results: A statistical approach. Biostat Biometrics Open Access J. 2020;6(1):112-119. Available from: https://doi.org/10.19080/BBOAJ.2020.06.555678
- Almeida S, Costa P. Statistical methods for hematological analyzer comparison. Stat Med. 2019;38(8):1465-1475. Available from: https://doi.org/10.1002/sim.8053
- Patel SP, Singh M. Performance assessment of hematological parameters using advanced analyzers. Int J Hematol Res. 2019;7(2):99-106. Available from: https://doi.org/10.19080/IJHR.2019.07.555710